Discover the value of integrated 3D medical device solutions
Explore our 3D printed medical device solutions
Why RICOH 3D for Healthcare
Powerful 3D printed medical device manufacturing
Work with our offsite production facility for delivery within a week, or let us come to you as a Ricoh-staffed managed service at your point of care.
No added work for your team - we handle everything
Avoid the cost and headaches of implementing 3D printing technologies and the regulatory hurdles of setting up your own medical device production.
Direct ordering - no workflow disruptions
Order from your familiar radiology viewer. Our PACS Print Gateway connects with various viewers so you can utilize your current workflow.
High quality and accuracy are built into the process
Ricoh's biomedical engineers segment DICOM files to your specifications. Robust quality control processes ensure accuracy of the model.
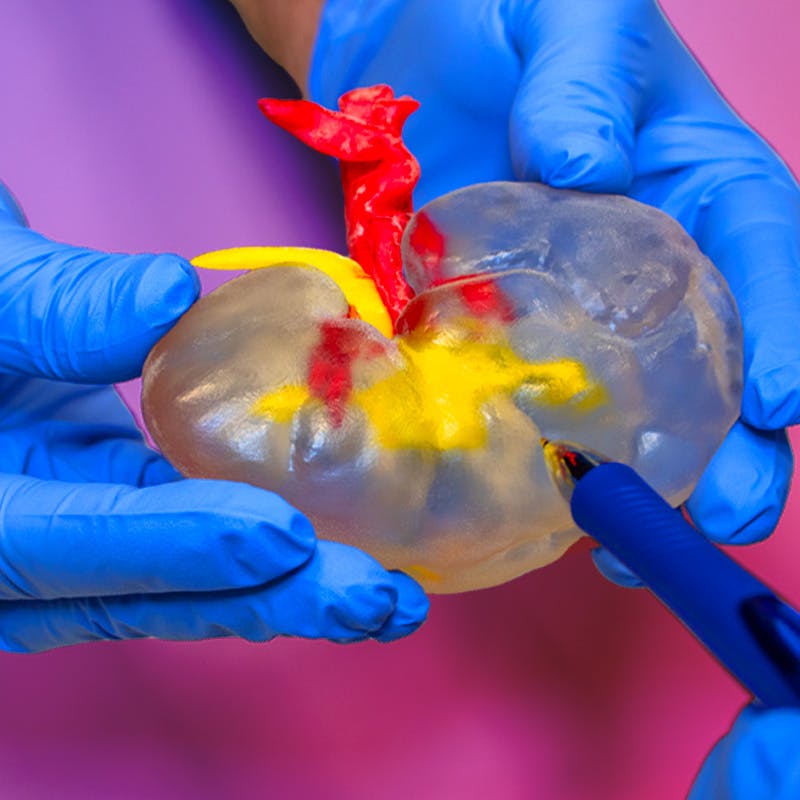
A mix of materials and colors help identification
Get the tactility you need to practice cuts effectively. Use colors to highlight vascular structures and tissue, or to identify anomalies such as tumors.
FDA-cleared for bolus and anatomic models
Our FDA 510(k) clearances allow us to produce patient-specific bolus and anatomic models.*
Customer stories
3D medical printing services offered
Partner manufacturing for MedTech
Prototype, manufacture, and commercialize medical devices with ISO 13485-certified facilities and expert engineers. Discover the possibilities of full-service 3D printing solutions, from design and development to packaging and distribution.
Video: Collaboration leads to clinical training phantom
Point-of-Care services for hospitals
Unlock potential with on-site 3D printing of FDA-cleared medical devices. Whether expanding an existing program or building a new one, accelerate innovation with a fully equipped and managed on-site studio— with minimal to no upfront costs required.
Video: See how one health system did it
Get a proof-of-value model for your organization
Additional insights on 3D printing in clinical settings
 Articles
ArticlesRicoh 3D anatomic models gain FDA 510(k) clearance
Ricoh’s FDA 510(k) clearance means healthcare providers don’t have to be experts in printing 3D models.
 Webinars
WebinarsSurgeons discuss using 3D-printed medical models
We’ve assembled a panel of expert surgeons who use 3D printed medical models to share why and how they’re using them.
 Articles
ArticlesMoving additive manufacturing forward with confidence
In part three of our series we share how to overcome the barriers to additive manufacturing and 3D printing.
- *FDA-cleared medical device for diagnostic use within craniomaxillofacial (CMF), orthopedic, cardiovascular, neurological, gastrointestinal, genitourinary, and breast applications.
- Ricoh USA, Inc.'s terms and conditions for anatomic models will apply to any proof-of-value request. Ricoh USA, Inc. reserves the right to reject requests for proof-of-value models in its sole discretion.